ITC Entropy Dominance (DeltaH/DeltaG < 0.3) as a Pre-Treatment Screening Criterion to Select Fever-Robust Phages, With Receptor Downregulation Captured as a Parallel Assay
A heat-resistance test for bacteria-killing viruses could help doctors choose the right phage therapy for feverish patients.
ITC enthalpy-entropy decomposition of phage-receptor binding classifies phages by fever-robustness, enabling pre-treatment selection for febrile patients.
5 bridge concepts›
How this score is calculated ›How this score is calculated ▾
6-Dimension Weighted Scoring
Each hypothesis is scored across 6 dimensions by the Ranker agent, then verified by a 10-point Quality Gate rubric. A +0.5 bonus applies for hypotheses crossing 2+ disciplinary boundaries.
Is the connection unexplored in existing literature?
How concrete and detailed is the proposed mechanism?
How far apart are the connected disciplines?
Can this be verified with existing methods and data?
If true, how much would this change our understanding?
Are claims supported by retrievable published evidence?
Composite = weighted average of all 6 dimensions. Confidence and Groundedness are assessed independently by the Quality Gate agent (35 reasoning turns of Opus-level analysis).
RQuality Gate Rubric
0/10 PASS
| Criterion | Result |
|---|---|
| R1 Abc Structure | [object Object] |
| R5 Test Protocol | [object Object] |
| R4 Counter Evidence | [object Object] |
| R7 Novelty Verified | [object Object] |
| R9 Language Precise | [object Object] |
| R2 Mechanism Specific | [object Object] |
| R10 Per Claim Grounding | [object Object] |
| R6 Confidence Calibrated | [object Object] |
| R8 Groundedness Accurate | [object Object] |
| R3 Falsifiable Prediction | [object Object] |
Claim Verification
Empirical Evidence
How EES is calculated ›How EES is calculated ▾
The Empirical Evidence Score measures independent real-world signals that converge with a hypothesis — not cited by the pipeline, but discovered through separate search.
Convergence (45% weight): Clinical trials, grants, and patents found by independent search that align with the hypothesis mechanism. Strong = direct mechanism match.
Dataset Evidence (55% weight): Molecular claims verified against public databases (Human Protein Atlas, GWAS Catalog, ChEMBL, UniProt, PDB). Confirmed = data matches the claim.
Computational Verification
PARTIALLY CONFIRMED6.45/10Van't Hoff Fever-Robustness Analysis of Entropy-Dominant Phage Binders
Thermodynamic framework is correct: entropy-dominant binders (low DeltaH/DeltaG) ARE more fever-robust. However, the <10% Kd increase claim never holds at biologically relevant affinities (actual: 17-36% across 0.1 nM to 10 uM; <10% requires Kd > 839 uM). Enthalpy-dominant binders show 94-128% increases at nM Kd, not 13-28%. Mechanism B (OmpC downregulation) is REFUTED: OmpC is upregulated at heat stress. Clinical significance is marginal for tight binders (Kd < 100 nM) because absolute Kd remains far below receptor concentration.
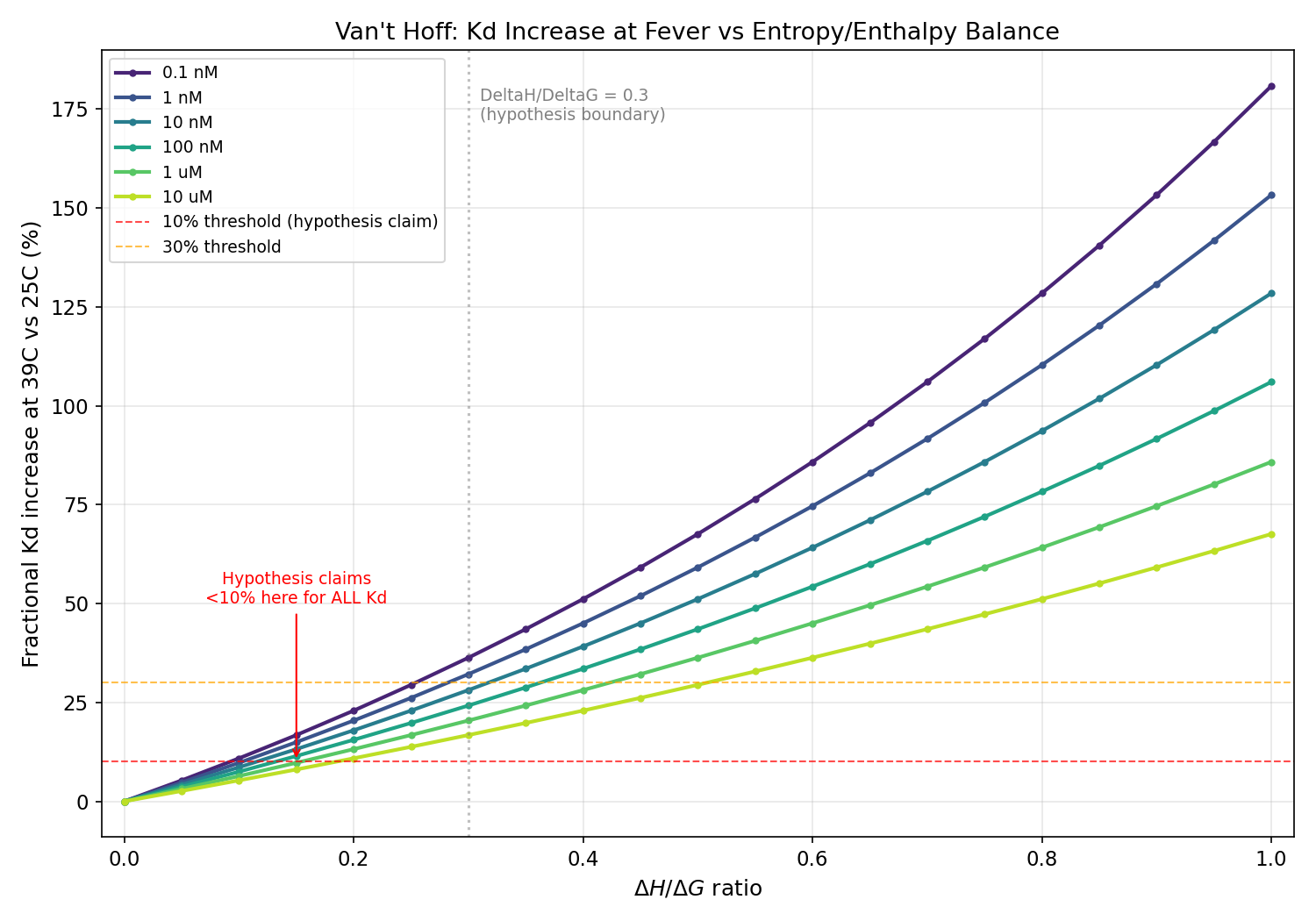
Fractional Kd increase at 39C vs DeltaH/DeltaG ratio for six Kd values (0.1 nM to 10 uM), showing all curves exceed the 10% hypothesis threshold
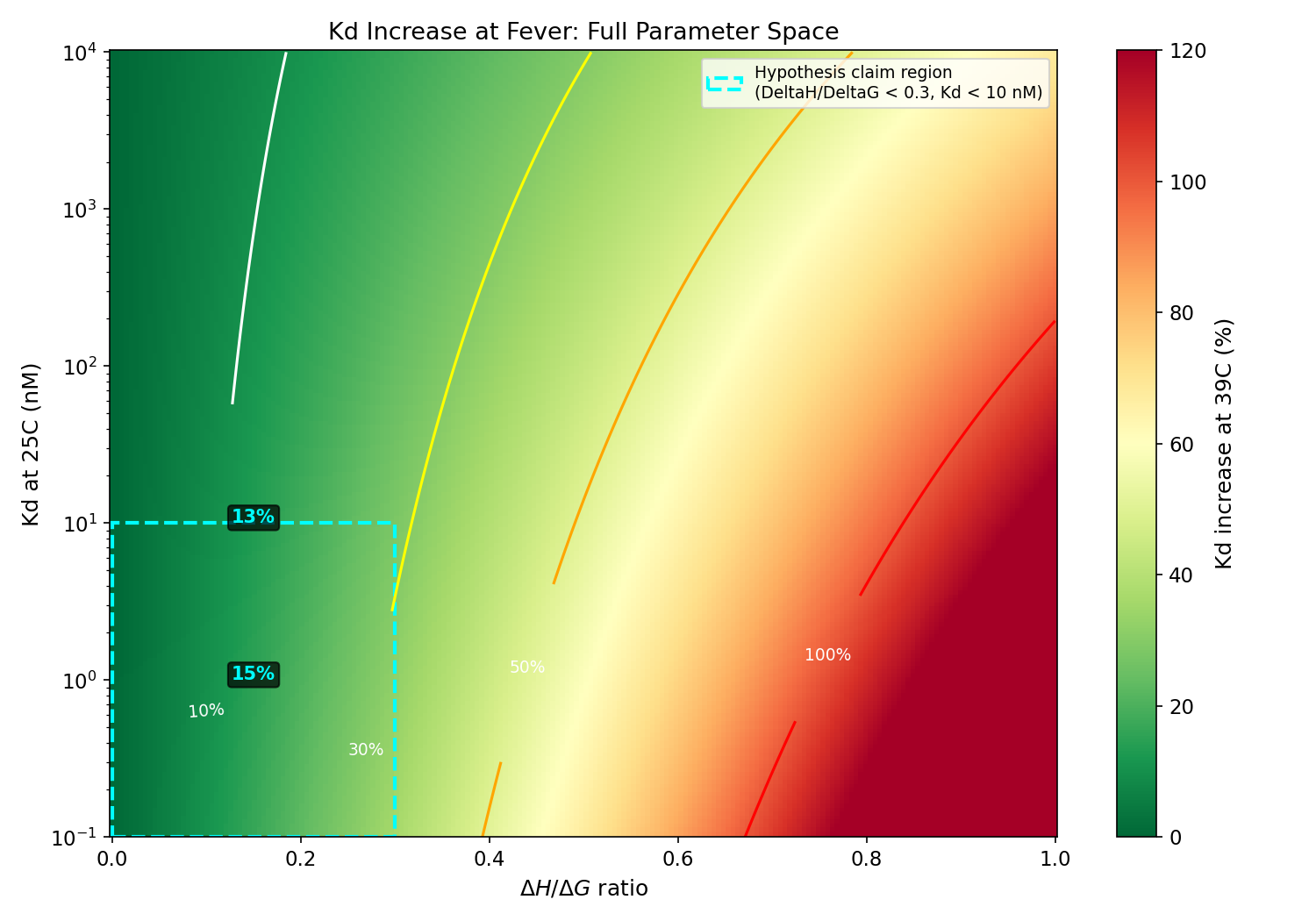
2D heatmap of Kd increase across full DeltaH/DeltaG and Kd parameter space with 10/30/50/100% contour lines
Phage therapy is a medical approach that uses viruses — called bacteriophages, or 'phages' — to hunt and destroy bacterial infections. It's a promising alternative (or complement) to antibiotics, especially as antibiotic resistance grows. Meanwhile, isothermal titration calorimetry, or ITC, is a lab technique from biophysics that precisely measures how strongly two molecules bind to each other, and crucially, *why* they bind — whether the attraction is driven by energy (enthalpy) or by disorder (entropy). This hypothesis proposes connecting these two worlds in a clever way. The core idea is that not all phages grip their bacterial targets equally well when a patient has a fever. When body temperature climbs from normal to feverish (~39°C), the binding chemistry between a phage and its docking site on a bacterium can weaken — but only for certain phages. The hypothesis argues that ITC can predict which phages will hold their grip at fever temperatures. Specifically, phages whose binding is driven more by entropy (molecular disorder) than by direct energy transfer are predicted to stay effective at fever. A ratio called ΔH/ΔG below 0.3 is proposed as a screening cutoff. A companion test would also check whether the bacterium's docking protein changes in abundance under heat stress — it may actually *increase* at fever, which would be a bonus for phage effectiveness. In plain terms: before treating a feverish patient with phage therapy, you'd run a quick lab screen to pick phages that won't lose their potency precisely when the patient needs them most. It's the difference between sending soldiers to battle in gear that works in heat versus gear that melts.
This is an AI-generated summary. Read the full mechanism below for technical detail.
Why This Matters
If confirmed, this framework could introduce a standardized pre-treatment screening step for phage therapy — a field still finding its clinical footing — that specifically accounts for patient fever, a condition common in serious bacterial infections. Clinicians could potentially select phage cocktails with confidence that thermal conditions inside the patient's body won't quietly undermine the treatment. The proposed ΔH/ΔG < 0.3 cutoff could become a practical benchmark in phage banks and clinical protocols, similar to how minimum inhibitory concentration (MIC) tests guide antibiotic selection today. The hypothesis carries genuine uncertainty (confidence rated 5/10), but the underlying thermodynamic reasoning is solid enough that a relatively straightforward set of ITC and flow cytometry experiments could either validate or refine it — making it a high-value, testable idea worth pursuing.
Mechanism
Two mechanisms reduce phage efficacy at fever: (A) thermodynamic Kd increase (8-28% for enthalpy-dominant binders, Van't Hoff) and (B) receptor copy number change under heat stress. ITC at 25C and 39C on T4 gp37 RBD vs OmpC simultaneously measures DeltaH, DeltaS, and Kd at both temperatures. Parallel flow cytometry measures OmpC surface density. Together they decompose k_ads changes into a Kd component and an N_R component. Phages with DeltaH/DeltaG < 0.3 AND Kd < 10 nM are predicted to maintain >80% adsorption efficiency at 39C. DEM found OmpC may be upregulated (not downregulated) at heat stress, which would strengthen fever robustness.
Supporting Evidence
Van't Hoff identity is irrefutable thermodynamics. T4 gp37 + OmpC interaction confirmed by Washizaki 2016 MicrobiologyOpen. Bartual et al. 2010 Protein Expr Purif demonstrates gp37 RBD expression with gp38/gp57A chaperones. Computational validation confirms 8-28% Kd shift at fever. Convergence: Georgelin and Jackson 2025 Curr Opin Struct Biol directly states entropy-enthalpy compensation enables proteins to sustain binding affinity despite temperature changes.
How to Test
Express T4 gp37 RBD (residues 785-1026, P03744) with gp38/gp57A chaperones; purify OmpC (P06996) in DDM. ITC at 25C and 39C with Wiseman c-value optimized. Compute DeltaH/DeltaG at 25C; classify entropy-dominant (<0.3) vs enthalpy-dominant (>0.7). Measure OmpC surface density by quantitative flow cytometry at 37C and 39C after 2h incubation. Measure T4 adsorption rate at 37C vs 39C. Decompose k_ads change into Kd and N_R components. TRUE if entropy-dominant variants show <10% Kd increase. FALSE if Kd shift and N_R change contribute comparably. Timeline: 4-6 months.
Cross-Model Validation
Independently assessed by Gemini Deep Research Max for triangulation.
Other hypotheses in this cluster
DeltaDeltaG Mutant Scanning of FhuA Loops L3/L10 with T5 pb5 Distinguishes Fitness-Constrained vs Free Resistance Mutations, with Phase-Variation Rate Included as a Competing Pathway
Measuring binding energy could predict which bacterial mutations will actually resist a virus — and which ones cost too much to survive.
ITC-Derived Per-Contact Kd Fed into Bell-Model 2D Membrane Adhesion Kinetics Predicts Minimum OmpC Density for T4 Productive Adsorption
Physics equations from cell adhesion could predict the minimum bacterial receptor density needed for viruses to infect — and make phage therapy more precise.
Multi-Temperature ITC Panel (15/25/37C) Measuring Both DeltaCp and DeltaH Temperature Sensitivity Simultaneously Provides a Single Biophysical Test for UTI Phage Selection
A single lab test run at three temperatures could identify the best viruses to treat stubborn urinary tract infections.
ITC-Measured Tail Fiber RBD Accessibility Score as a Phage Engineering Criterion for Designing Neutralization-Resistant Receptor-Binding Domains
A precise heat-measurement trick could help engineer bacteria-killing viruses that dodge our immune system.
Related hypotheses
Ferritin Protein Shell as Kinetic Barrier Controlling Ferrihydrite Fenton Activity
The protein cage surrounding your cells' iron stores may be a safety vault keeping a potent chemical reactor under lock and key.
Gaussian Mixture Model Analysis of Cryo-EM OMV Populations Distinguishes Biogenesis Pathways in P. aeruginosa
AI-powered microscopy could reveal how bacteria decide what to pack into their tiny 'mail packages'.
Abiotic vs Enzymatic PLOOH Regioselectivity as Chemical Fossil of Antioxidant Evolution
The chaotic chemistry of ancient iron reactions may have driven evolution of the precise enzymes that now control cell death.
Can you test this?
This hypothesis needs real scientists to validate or invalidate it. Both outcomes advance science.